The pharmaceutical grade twin-doors washer is a new type of equipment for pharmaceutical grade aseptic garment cleaning designed according to the basic principles of Euramerican aseptic garment cleaning equipment, the fabric properties of aseptic garments, and their cleaning technological requirements in B, C, and D grade sterile area of pharmaceutical production. The combination of Euramerican aseptic garment washer as a basis with new version GMP in China and FDA in the US makes this equipment ensure that cleanliness and asepsis degree of the cleaned garments meet the requirements of new version GMP, CGMP, FDA, E/DMF, and COS, etc. Being the only pharmaceutical grade aseptic garment cleaning equipment that meets aforementioned requirements at present in our country, it is indispensable to modern pharmaceutical enterprises. This equipment, filling the gap in garment cleaning and sterilization equipment for pharmaceutical industry which has hitherto existed in China, is completely different from the industrial and domestic washers that were widely used in pharmaceutical enterprises. It realized the establishment of a totally automatic work station for aseptic garment processing and its integration into cleanroom aseptic garment management system, eradicating cross-contamination (garments before and after cleaning are processed in the same grade sterile area) and other problems disobeying GMP requirements that occur when using a single-door industrial or domestic washer.
Besides overcoming the problems of using ordinary domestic washers or traditional industrial washers, the equipment enables garment cleanliness degree to be tested by a Helmke Drum which is used worldwide. Similar washers have already been extensively adopted by pharmaceutical enterprises in Europe and America.
Other characteristics include:
(1) Simple technological layout and planimetric layout due to simplified garment processing.
(2) Easy installation, good sealing, and convenient layout due to twin-doors container-embedded installation.
(3) Reduced operating cost due to saving space, energy consumption, and personnel.
(4) Complete records, high traceability and reproducibility, and mature technical conditions for validation and re-validation. 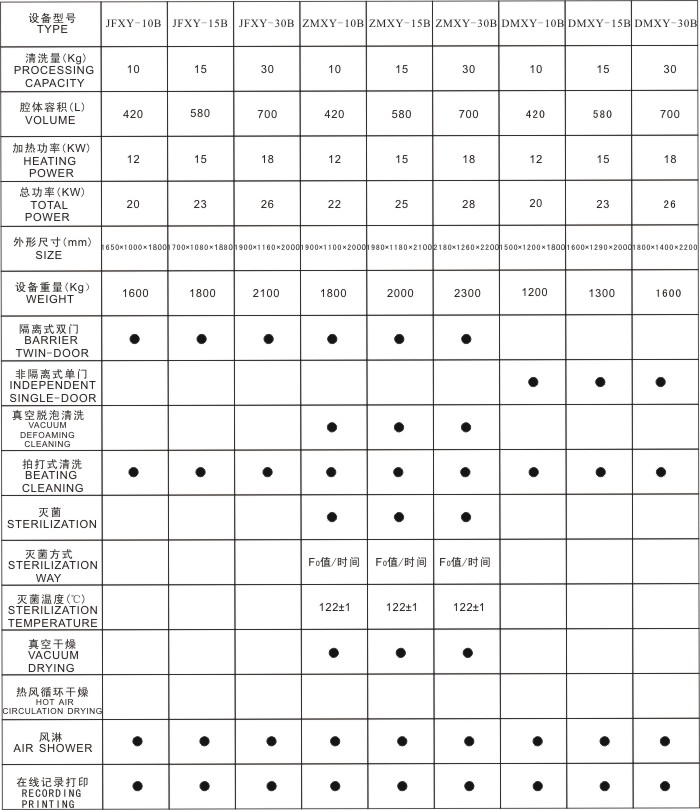
|
